Selected Publications
Droplet Squeeze Microfluidic Platform for Generating Extracellular Vesicle Hybrids for Drug Delivery
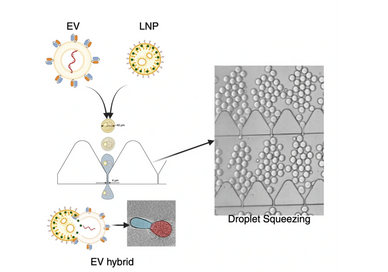
Extracellular vesicles (EVs) are emerging as versatile drug delivery systems due to their intrinsic biocompatibility and targeting capabilities. However, EV integrity and efficient drug loading challenges hinder their clinical translation. To address these limitations, hybrid systems integrating lipid nanoparticles (LNPs) with EVs have gained attention for their potential in targeted and combinatorial drug delivery. This study presents a robust microfluidic approach for the scalable generation of drug-loaded EV-LNP hybrids (EV hybrids). The method facilitates controlled fusion between EVs and LNPs by utilizing a droplet-mediated squeezing mechanism. Lipid composition and microfluidic parameters are optimized for the fusion of EVs and LNPs and determined physicochemical and functional characterizations of the EV hybrids. In vitro studies demonstrate that EV hybrids exhibit enhanced targeting efficiency. Moreover, small-molecule therapeutics are successfully encapsulated within EV hybrids, significantly improving cytotoxic efficacy against melanoma in 2D and 3D culture models compared to drug-loaded EVs or LNPs alone. The work introduces a scalable, minimally disruptive microfluidic platform for engineering EV hybrids, offering a promising strategy to advance precision nanomedicine.
Enhancing Chimeric Antigen Receptor T-Cell Generation via Microfluidic Mechanoporation and Lipid Nan
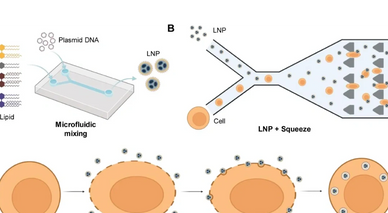
Chimeric antigen receptor (CAR)-T cell therapy has revolutionized cancer treatment by engineering patients' T cells to specifically target cancer cells. Traditional CAR-T cell manufacturing methods use viral transduction to integrate CAR genes into T cells, but this can cause severe side effects and immune reactions and is costly. To overcome these challenges, non-viral methods, such as plasmid DNA (pDNA) transfection, are being explored. Here, a high-throughput intracellular delivery platform that integrates microfluidic mechanoporation with lipid nanoparticle (LNP)-based delivery, LNP + Squeeze, is introduced. This system enhances pDNA transfection efficiency in T cells while maintaining cell viability compared to other non-viral transfection methods like electroporation. This platform successfully engineers CAR-T cells using primary human T cells with a high transfection efficiency and demonstrates potent cytotoxicity against melanoma cells. This approach offers a promising, cost-effective, and scalable alternative to viral methods, potentially improving the accessibility and efficacy of CAR-T cell therapies.
Vertically Aligned Nanowires for Longitudinal Intracellular Sampling
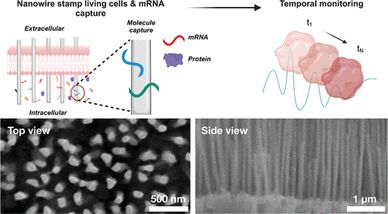
Cells are diverse systems with unique molecular profiles that support vital functions, such as energy production and nutrient absorption. Advances in omics have provided valuable insights into these cellular processes, but many of these tools rely on cell lysis, limiting the ability to track dynamic changes over time. To overcome this, methods for longitudinal profiling of living cells have emerged; however, challenges such as low throughput and genetic manipulation still need to be addressed. Nanomaterials, particularly nanowires, offer a promising solution due to their size, high aspect ratios, low cost, simplicity, and potential for high-throughput manufacturing. Here, we present a nanowire-based platform for longitudinal mRNA profiling in living cells using vertically aligned nickel nanowire arrays for efficient mRNA extraction with minimal cellular disruption. We demonstrate its ability to track enhanced green fluorescent protein expression and transcriptomic changes from drug responses in the same cells over time, showcasing the platform's potential for dynamic cellular analysis
Squeezable Hydrogel Microparticles for Single Extracellular Vesicle Protein Profiling
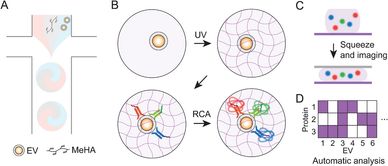
Extracellular vesicles (EVs) are promising for molecular diagnostics, but current analyses are limited by the rarity and compositional heterogeneity of EV protein expression. Therefore, single EV profiling methods require high sensitivity, multiplexing, and throughput to address these issues. Here a single EV analysis technique that utilizes squeezable methacrylated hyaluronic acid hydrogel microparticles (MHPs) is described as a scaffold to immobilize EVs and perform an integrated rolling circle amplification (RCA) assay for an ultra-sensitive and multiplex analysis of single EV proteins. EVs are prepared into MHPs in a high-throughput manner with droplet microfluidics and optimally labeled with antibody-oligonucleotide conjugates in MHPs without steric limitations. By designing MHPs with high compressibility, single EV protein signals are amplified as RCA products that can be aligned on the same plane by physically squeezing MHPs and visualized with low magnification. This method provides a simple and scalable single EV imaging analysis pipeline for identifying multiplex marker expression patterns from single EVs. For validation, the single EV heterogeneity of highly expressed cancer cell markers is profiled across different cancer cell lines. These findings exemplify squeezable MHPs as a robust platform with high sensitivity, multiplexing, and scalability for resolving single EV heterogeneity and advancing molecular assay technologies.
Immunomodulation in age-related disorders and nanotechnology interventions
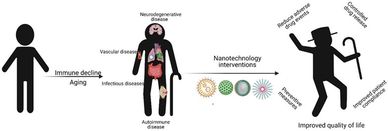
Recently, the aging population has increased exponentially around the globe bringing more challenges to improve quality of life in those populations while reducing the economic burden on healthcare systems. Aging is associated with changes in the immune system culminating in detrimental effects such as immune dysfunction, immunosenescence, and chronic inflammation. Age-related decline of immune functions is associated with various pathologies including cardiovascular, autoimmune, neurodegenerative, and infectious diseases to name a few. Conventional treatment addresses the onset of age-related diseases by early detection of risk factors, administration of vaccines as preventive care, immunomodulatory treatment, and other dietary supplements. However, these approaches often come with systemic side-effects, low bioavailability of therapeutic agents, and poor outcomes seen in the elderly. Recent innovations in nanotechnology have led to the development of novel biomaterials/nanomaterials, which explore targeted drug delivery and immunomodulatory interactions in vivo. Current nanotechnology-based immunomodulatory approaches that have the potential to be used as therapeutic interventions for some prominent age-related diseases are discussed here. Finally, we explore challenges and future aspects of nanotechnology in the treatments of age-related disorders to improve quality of life in the elderly.
Enhancing Chimeric Antigen Receptor T-Cell Generation via Microfluidic Mechanoporation and Lipid Nan

Chimeric antigen receptor (CAR)-T cell therapy has revolutionized cancer treatment by engineering patients' T cells to specifically target cancer cells. Traditional CAR-T cell manufacturing methods use viral transduction to integrate CAR genes into T cells, but this can cause severe side effects and immune reactions and is costly. To overcome these challenges, non-viral methods, such as plasmid DNA (pDNA) transfection, are being explored. Here, a high-throughput intracellular delivery platform that integrates microfluidic mechanoporation with lipid nanoparticle (LNP)-based delivery, LNP + Squeeze, is introduced. This system enhances pDNA transfection efficiency in T cells while maintaining cell viability compared to other non-viral transfection methods like electroporation. This platform successfully engineers CAR-T cells using primary human T cells with a high transfection efficiency and demonstrates potent cytotoxicity against melanoma cells. This approach offers a promising, cost-effective, and scalable alternative to viral methods, potentially improving the accessibility and efficacy of CAR-T cell therapies.
Nanocomposites to treat SARS-CoV-2
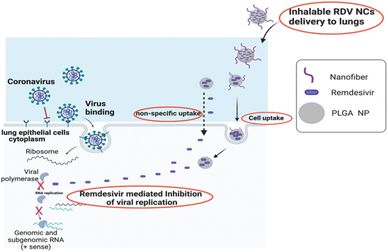
To develop an inhalable drug delivery system, we synthesized poly (lactic-co-glycolic acid) nanoparticles with Remdesivir (RDV NPs) as an antiviral agent against SARS-CoV-2 replication and formulated Remdesivir-loaded nanocomposites (RDV NCs) via coating of RDV NPs with novel supramolecular cell-penetrating peptide nanofibers (NFs) to enhance cellular uptake and intracellular drug delivery. RDV NPs and RDV NCs were characterized using variou techniques, including Transmission Electron Microscopy (TEM), Dynamic Light Scattering (DLS), and fluorescent microscopy. The cytotoxicity of RDV NCs was assessed in Vero E6 cells and primary human lung epithelial cells, with no significant cytotoxicity observed up to 1000 μg mL−1 and 48 h. RDV NCs were spherically shaped with a size range of 200–300 nm and a zeta potential of ∼+31 mV as well as indicating the presence of coated nanofibers. Reverse Transcription-quantitative Polymerase Chain Reaction (RT-qPCR), immunofluorescence and plaque assays of SARS-CoV-2 infected Vero E6 treated with RDV NCs showed significantly higher antiviral activities compared to those of free drug and uncoated RDV NPs. RDV NCs exhibited high antiviral activity against SARS-CoV-2, and the nanocomposite platform has the potential to be developed into an inhalable drug delivery system for other viral infections in the lungs.
Targeted Chemotherapy- Cell membrane coated nanoparticles

Cell membrane-derived nanoparticles (NPs) have recently gained popularity due to their desirable features in drug delivery such as mimicking properties of native cells, impeding systemic clearance, and altering foreign body responses. Besides NP technology, adoptive immunotherapy has emerged due to its promise in cancer specificity and therapeutic efficacy. In this research, we developed a biomimetic drug carrier based on chimeric antigen receptor (CAR) transduced T-cell membranes. For that purpose, anti-HER2 CAR-T cells were engineered via lentiviral transduction of anti-HER2 CAR coding lentiviral plasmids. Anti-HER2 CAR-T cells were characterized by their specific activities against the HER2 antigen and used for cell membrane extraction. Anti-cancer drug Cisplatin-loaded poly (D, l-lactide-co-glycolic acid) (PLGA) NPs were coated with anti-human epidermal growth factor receptor 2 (HER2)-specific CAR engineered T-cell membranes. Anti-HER2 CAR-T-cell membrane-coated PLGA NPs (CAR-T-MNPs) were characterized and confirmed via fluorescent microscopy and flow cytometry. Membrane-coated NPs showed a sustained drug release over the course of 21 days in physiological conditions. Cisplatin-loaded CAR-T-MNPs also inhibited the growth of multiple HER2+ cancer cells in vitro. In addition, in vitro uptake studies revealed that CAR-T-MNPs showed an increased uptake by A549 cells. These results were also confirmed via in vivo biodistribution and therapeutic studies using a subcutaneous lung cancer model in nude mice. CAR-T-MNPs localized preferentially at tumor areas compared to those of other studied groups and consisted of a significant reduction in tumor growth in tumor-bearing mice. In Conclusion, the new CAR modified cell membrane-coated NP drug-delivery platform has demonstrated its efficacy both in vitro and in vivo. Therefore, CAR engineered membrane-coated NP system could be a promising cell-mimicking drug carrier that could improve therapeutic outcomes of lung cancer treatments.
Gene Therapy- In vivo Evaluation of Non-viral NICD Plasmid-Loaded PLGA Nanoparticles in Developing Z
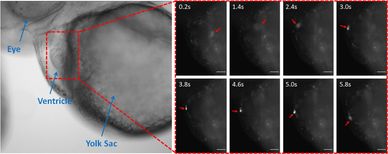
In the era of the advanced nanomaterials, use of nanoparticles has been highlighted in biomedical research. However, the demonstration of DNA plasmid delivery with nanoparticles for in vivo gene delivery experiments must be carefully tested due to many possible issues, including toxicity. The purpose of the current study was to deliver a Notch Intracellular Domain (NICD)-encoded plasmid via poly(lactic-co-glycolic acid) (PLGA) nanoparticles and to investigate the toxic environmental side effects for an in vivo experiment. In addition, we demonstrated the target delivery to the endothelium, including the endocardial layer, which is challenging to manipulate gene expression for cardiac functions due to the beating heart and rapid blood pumping. For this study, we used a zebrafish animal model and exposed it to nanoparticles at varying concentrations to observe for specific malformations over time for toxic effects of PLGA nanoparticles as a delivery vehicle. Our nanoparticles caused significantly less malformations than the positive control, ZnO nanoparticles. Additionally, the NICD plasmid was successfully delivered by PLGA nanoparticles and significantly increased Notch signaling related genes. Furthermore, our image based deep-learning analysis approach evaluated that the antibody conjugated nanoparticles were successfully bound to the endocardium to overexpress Notch related genes and improve cardiac function such as ejection fraction, fractional shortening, and cardiac output. This research demonstrates that PLGA nanoparticle-mediated target delivery to upregulate Notch related genes which can be a potential therapeutic approach with minimum toxic effects.
Nanocomposites for enhanced intracellular drug delivery

Effective drug delivery to pulmonary sites will benefit from the design and synthesis of novel drug delivery systems that can overcome various tissue and cellular barriers. Cell penetrating peptides (CPPs) have shown promise for intracellular delivery of various imaging probes and therapeutics. Although CPPs improve delivery efficacy to a certain extent, they still lack the scope of engineering to improve the payload capacity and protect the payload from the physiological environment in drug delivery applications. Inspired by recent advances of CPPs and CPP-functionalized nanoparticles, in this work, we demonstrate a novel nanocomposite consisting of fiber-forming supramolecular CPPs that are coated onto polylactic-glycolic acid (PLGA) nanoparticles to enhance pulmonary drug delivery. These nanocomposites show a threefold higher intracellular delivery of nanoparticles in various cells including primary lung epithelial cells, macrophages, and a 10-fold increase in endothelial cells compared to naked PLGA nanoparticles or a twofold increase compared to nanoparticles modified with traditional monomeric CPPs. Cell uptake studies suggest that nanocomposites likely enter cells through mixed macropinocytosis and passive energy-independent mechanisms, which is followed by endosomal escape within 24 h. Nanocomposites also showed potent mucus permeation. More importantly, freeze-drying and nebulizing formulated nanocomposite powder did not affect their physiochemical and biological activity, which further highlights the translative potential for use as a stable drug carrier for pulmonary drug delivery. We expect nanocomposites based on peptide nanofibers, and PLGA nanoparticles can be custom designed to encapsulate and deliver a wide range of therapeutics including nucleic acids, proteins, and small-molecule drugs when employed in inhalable systems to treat various pulmonary diseases.
Gene Therapy- Notch Intracellular Domain Plasmid Delivery via Poly(Lactic-Co-Glycolic Acid) Nanopart

Notch signaling is a highly conserved signaling system that is required for embryonic development and regeneration of organs. When the signal is lost, maldevelopment occurs and leads to a lethal state. Delivering exogenous genetic materials encoding Notch into cells can reestablish downstream signaling and rescue cellular functions. In this study, we utilized the negatively charged and FDA approved polymer poly(lactic-co-glycolic acid) to encapsulate Notch Intracellular Domain-containing plasmid in nanoparticles. We show that primary human umbilical vein endothelial cells (HUVECs) readily uptake the nanoparticles with and without specific antibody targets. We demonstrated that our nanoparticles are non-toxic, stable over time, and compatible with blood. We further demonstrated that HUVECs could be successfully transfected with these nanoparticles in static and dynamic environments. Lastly, we elucidated that these nanoparticles could upregulate the downstream genes of Notch signaling, indicating that the payload was viable and successfully altered the genetic downstream effects.
Anti-cancer bioactivity of sweet basil leaf derived extracellular vesicles on pancreatic cancer cell
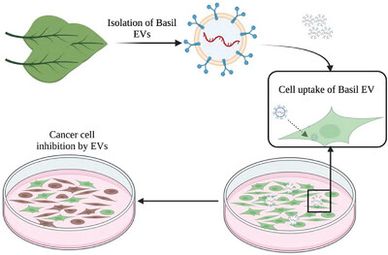
Most living organisms secrete tiny lipid bilayer particles encapsulating various biomolecular entities, including nucleic acids and proteins. These secreted extracellular vesicles (EVs) are shown to aid in communication between cells and their environment. EVs are mainly involved in the signalling and manipulation of physiological processes. Plant EVs display similar functional activity as seen in mammalian EVs. Medicinal plants have many bioactive constituents with potential applications in cancer treatment. Particularly, Basil (Ocimum basilicum), has wide therapeutic properties including anti-inflammatory, anti-cancer, and anti-infection, among others. In this study, we focused on using EVs purified from Apoplast Washing Fluid (AWF) of Basil plant leaves as a biological therapeutic agent against cancer. Characterization of Basil EVs revealed a size range of 100–250 nm, which were later assessed for their cell uptake and apoptosis inducing abilities in pancreatic cancer cells. Basil plant EVs (BasEVs) showed a significant cytotoxic effect on pancreatic cancer cell line MIA PaCa-2 at a concentration of 80 and 160 μg/mL in cell viability, as well as clonogenic assays. Similarly, RT-PCR and western blot analysis has shown up regulation in apoptotic gene and protein expression of Bax, respectively, in BasEV treatment groups compared to untreated controls of MIA PaCa-2. Overall, our results suggest that EVs from basil plants have potent anti-cancer effects in pancreatic cancer cells and can serve as a drug delivery system, demanding an investigation into the therapeutic potential of other medicinal plant EVs
Book Chapters

Droplet Microfluidics
Over the last few decades, the discipline of microfluidics has grown in importance in the medical, engineering, information technology, and energy fields. One of the more popular subdisciplines that have emerged in this area, though, is droplet microfluidics. Droplet microfluidics entails the creation of tens of thousands of monodisperse droplets for performing countless amount of reactions and high-throughput analyses. Because their parameters are easily adjustable, a plethora of droplets with different compositions and structures can be formed and investigated. In this chapter, we aim to provide a comprehensive overview of the fundamentals, methodologies, and applications behind droplet production and its significance in the microfluidic field. The highlighted material will provide a better understanding of droplet microfluidics’ past, present, and future.
Presentations
2025 Presented "Squeezing droplets make better cancer nanomedicine". at Start Talking Science at Science history institute, Philadelphia, USA
2024 Invited Speaker at Center for Innovation & Precision Dentistry, “Developing Targeted Cancer Treatments with Microfluidic-Engineered Natural Vesicles”,
2024 “Gene Therapy using Nanoparticles” Invited Guest lecture University of Texas at Arlington
2022 “Drug Delivery using nanocomposites for treatment of COVID-19”, Oral Presentation at BMES 2022 Annual Meeting.
2022 “Nanocomposites for drug delivery to treat MRSA bacterial infection “, Oral presentation at BMES 2022 Annual Meeting
2022 “Assessing Local Delivery Capability of a Novel Septal Ablation System via an Ex Vivo Pig Heart Model”, Poster presentation at Summer Biomechanics, Bioengineering, and Biotransport Conference, Maryland.
2022 “Nanoparticle targeting E-CAM1 in atherosclerosis from age0related cardiovascular disease”, Poster presentation at BMES 2022 Annual Meeting.
2022 “Development of Injectable nanocomposite hydrogels for stimuli responsive pain medicine delivery” University of Texas at Arlington Innovation Day.
2022 “Biomechanical and Microstructural Characterization of Human Healthy and hypertrophic Septal Tissues” Poster presented at Summer Biomechanics, Bioengineering, and Biotransport Conference.
2021 “Development of Lipid-based nanoparticles to treat SARS-CoV-2 lung infections in vitro” Poster presentation at Social Connection Conference, Texas.
2020 “Nanoparticle-mediated controlled myocardial Delivery for hypertrophic cardiomyopathy” poster presentation at BMES 2020 Annual Meeting. (NIH R15 $433,000 awarded)
2020 “Invited Expert Opinion article on media for future of Biotechnology”, Collaborative Drug Discovery, USA.
2020 “Development of nanoparticles to treat lung infections via delivery of antibiotics” Poster presentation at BMES 2020 Annual Meeting.

This website uses cookies.
We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.